DTLs are generally submitted to the Clinical Investigator (CI) for approval by the DTL Administrator (DTLA), although for brand new DTLs, the submitter is not required to be a DTLA. Furthermore, DTLs that are due for annual approval aren’t submitted, but are automatically provided to the CI for signature.
Three types of submit actions are described in this section:
- Submitting a Single Initiated DTL for Approval
- Submitting an Updated DTL for Approval
- Submitting Multiple Initiated DTLs for Approval
Submitting a Single Initiated DTL for Approval
To submit a new/initiated site DTL for CI sign-off, the site must:
- Have a current IRB approval on file with the CTSU. If no IRB approval is on file for the site, a status reason of “No IRB Approval on file for this site” will appear in the status reason column for the following task assignments: Clinical Investigator, Enrolling Person/Treating Investigator, and Consenting Person.
- Ensure all required tasks are assigned to satisfy the minimum requirements.
When all required task assignments are made and the site’s IRB approval is on file, the Submit DTL button will be enabled (see Figure 13) and can be clicked to submit the DTL for CI sign-off; a confirmation popup will appear. Once the system processes the request, the DTL’s status will update to Awaiting CI Approval. Note that the CI will not receive an email about the new site DTL unless the DTLA uses the Notify CI functionality (see next section; the CTSU recommends that this feature be reserved for situations where the CI signature is required urgently). Otherwise, the new site DTL will be available on the CI’s DTL Approvals screen and will be part of the Bi-Monthly Signing Notification email.
Figure 13: Enabled Submit DTL button.
If the assigned CI or DTLA needs to make changes to a DTL the first time it is in an Awaiting CI Approval status, they may revert the DTL to an Initiated state, make the necessary updates, and then re-submit the DTL for CI approval. To revert the DTL, locate the DTL under the Site DTL Browser and select the Revert  icon above the Task Assignments grid on the Manage Site DTL screen (see Figure 14).
icon above the Task Assignments grid on the Manage Site DTL screen (see Figure 14).
Figure 14: Revert DTL icon.
A popup will appear to confirm that you would like to revert the DTL to an Initiated status. Select Confirm to finish reverting the DTL status. A message will display if the DTL was successfully reverted, and the DTL status will change back to Initiated.
Submitting an Updated DTL for Approval
When a site DTL has changes that require CI signature, it will automatically be made available on the CI’s DTL Approvals screen and will be part of the Bi-Monthly Signing Notification email. If there is an urgent need for the CI to sign the updated DTL, the assigned DTLA can send an email notification to the CI that the updated DTL is available for approval. A Notify CI button should display above the Task Assignments grid on the Manage Site DTL screen (see Figure 15).

Figure 15: Enabled Notify CI button.
The button will display if there is valid reason for signing the DTL:
- The DTL is in Awaiting CI Approval, Approved, or Unapproved status.
- The current user has an Active, Awaiting CI Approval, or Pending DTLA task assignment on the DTL.
- There is a valid reason for signing the Site DTL.
- Reasons include when the site DTL has changes ready to be approved and there is at least one task on the DTL where the task status is in Awaiting CI Approval.
- Note that not all task assignments require a CI signature to be activated. See the DTL Master Task List and the Clinical Investigator (CI) Signature Requirement column for information on which tasks do not require signature.
On click of the button, a confirmation popup should appear (see Figure 16). Select Send Email to send an email to the CI. There are no restrictions on the number of emails a DTLA can send for a given DTL, but as noted previously, the CTSU recommends using this feature judiciously.
Figure 16: Confirmation request popup with Send Email button.
Submitting Multiple Initiated DTLs for Approval
The bulk submit DTLs for approval operation allows users to submit multiple initiated site DTLs to the CI(s) for approval. Any rostered site staff member can submit initiated DTLs for approval, they do not need to be a CI or DTLA.
Site users can access the Submit Initiated DTLs to CI for Approval screen by selecting Site DTL > Submit for Approval in the menu or via the Submit DTLs for Approval Quick Link in the Dashboard (see Figures 17 and 18).
Figure 17: Submit for Approval menu item.
Figure 18: Submit DTLs for Approval Quick Link.
If the user does not have any DTLs available to be submitted, they will receive a message stating this fact.
If they do have DTLs available for submission, the resulting screen contains two sections, DTL Submit Summary and the DTLs in Initiated Status grid.
DTL Submit Summary
The DTL Submit Summary section displays if the user has at least one DTL to submit for approval. The summary will list the number of DTLs that are selected out of the total number possible in the grid, as well as a Submit button (see Figure 19).
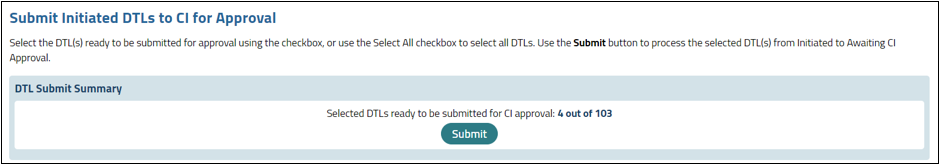
Figure 19: DTL Submit Summary.
DTLs in Initiated Status Grid
The DTLs in Initiated Status section includes the following information (see Figure 20):
- Select All DTL(s) - when this checkbox is selected, the checkbox in every row of the grid will be checked. The total number of DTLs will display in the DTL Submit Summary.
- Each row in the grid has a checkbox allowing you to select or unselect individual DTLs.
- Site - the CTEP ID for each site relevant to an Initiated DTL. An i button
 next to the site provides more information on the institution.
next to the site provides more information on the institution.
- LPO - the Lead Protocol Organization (LPO) associated with the DTL.
- Protocol - the protocol relevant to the DTL. An i button
 next to the protocol provides more information on the protocol.
next to the protocol provides more information on the protocol.
- Protocol Status - the status of the protocol.
- Assigned CI - the name of the CI assigned on the DTL. An i button
 next to the assigned CI provides more information on the person.
next to the assigned CI provides more information on the person.
- Updated Date - the date when the DTL was last updated.
- Actions - a View DTL icon
 will display, allowing the user to review information on the DTL in the Manage Site DTL screen in a new browser tab.
will display, allowing the user to review information on the DTL in the Manage Site DTL screen in a new browser tab.
The ability to sort, filter, and export grid data is available; filters are hidden by default but can be displayed using the funnel icon.
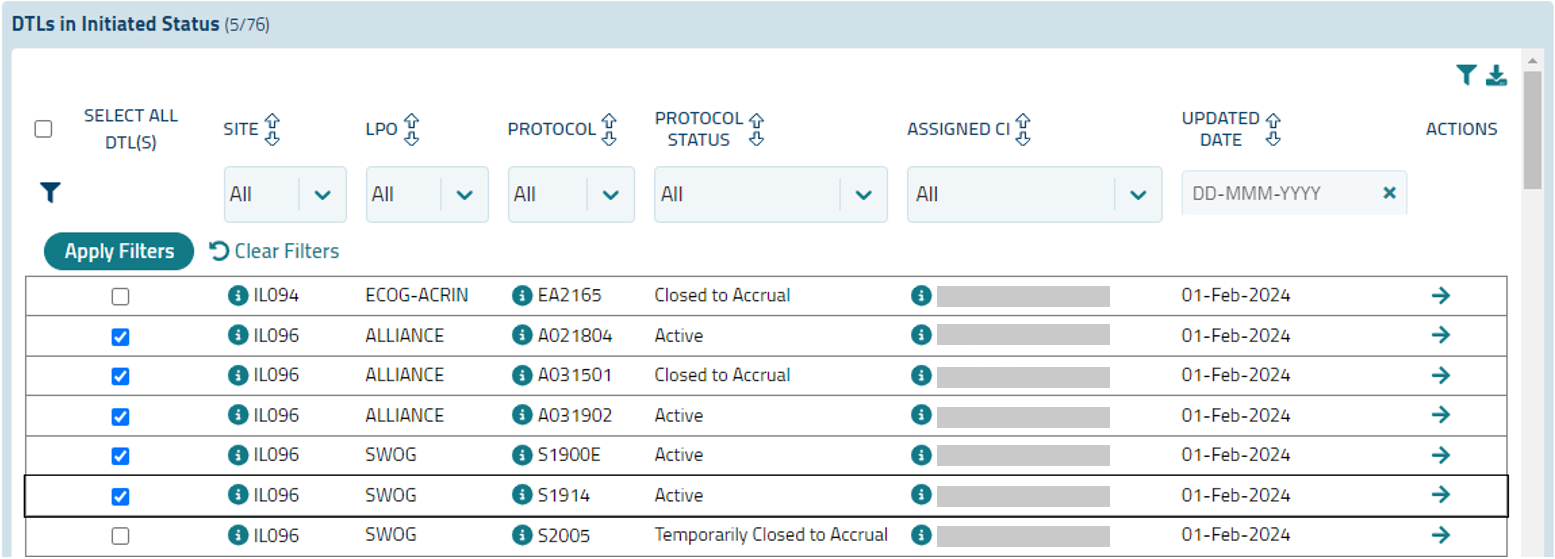
Figure 20: DTLs in Initiated Status grid.
Submit Process
A Submit button is visible in the DTL Submit Summary section. The button is enabled if at least one DTL in the DTLs in Initiated Status grid has been selected.
When the button is clicked, a popup appears. A message appears in the popup, asking you to confirm whether you want to submit the DTLs (# selected from the grid) to the CI(s) for approval (see Figure 21).
Figure 21: Submit DTLs confirmation request.
Click on the Confirm button and the request will be processed. You will then be taken to a Confirmation screen (see Figure 22).
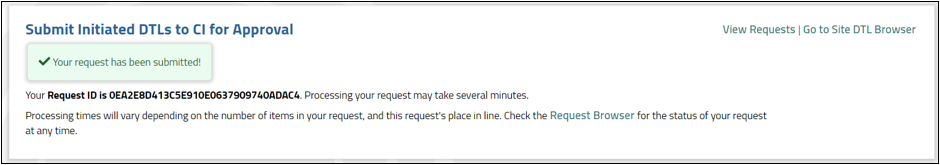
Figure 22: Submit Initiated DTLs to CI for Approval confirmation screen.
- Click the View Requests link to navigate to the Request Browser. All requests will display in the grid.
- Click the Go to Site DTL Browser link to navigate to the Site DTL Browser page. Note that if you return to the Site DTL Browser, you will see the newly submitted DTLs, but they will be locked until all processing is complete. At that point, the locks will be removed and the DTLs will be in the Awaiting CI Approval status.
- Click the Request Browser link within the body of the message to view the progress of this individual request, as well as details about the request.
- Note: See the Help Topic Request Browser Overview for more information about the Request Browser.